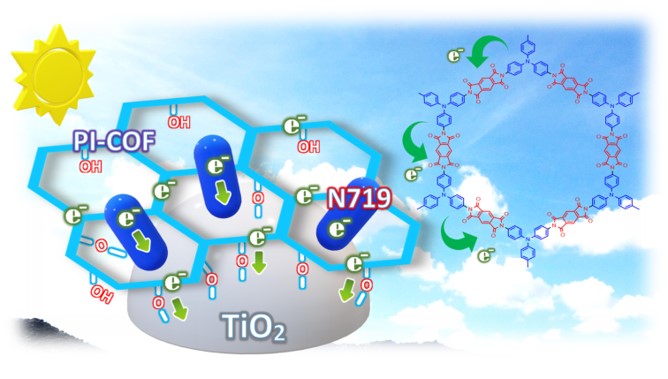
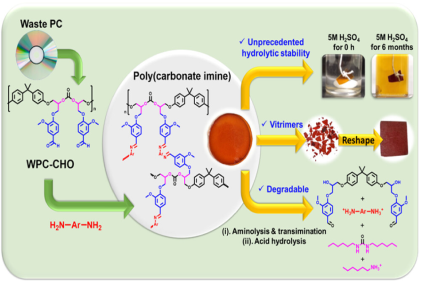
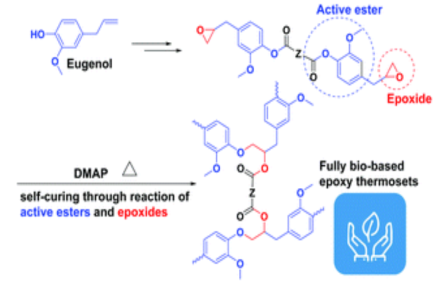
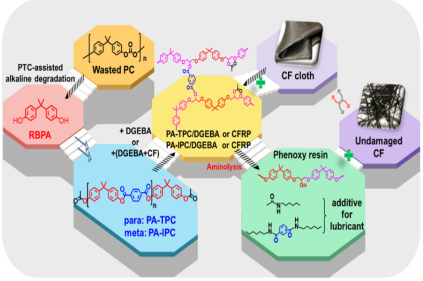
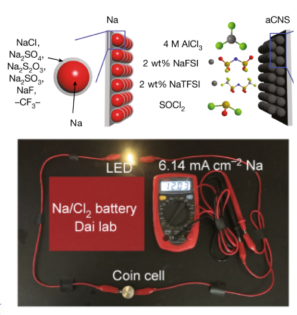
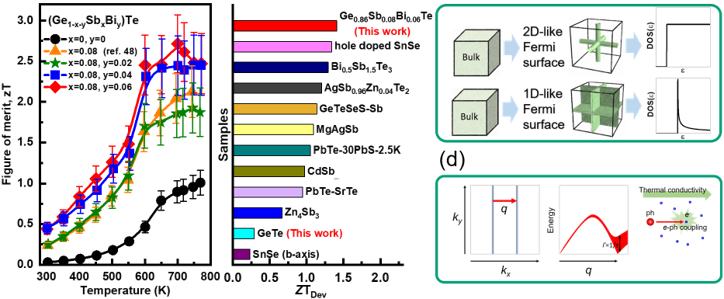
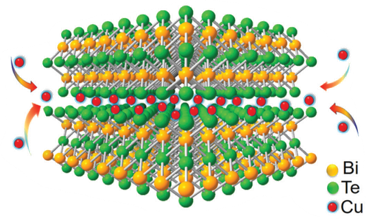
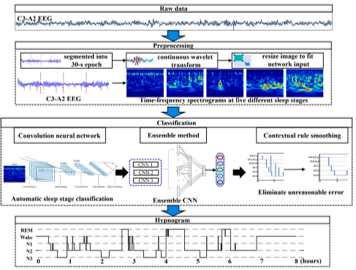
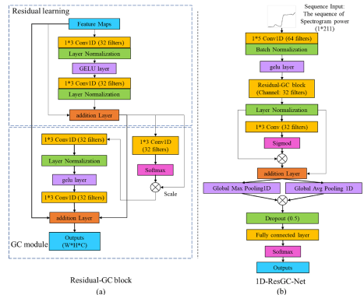
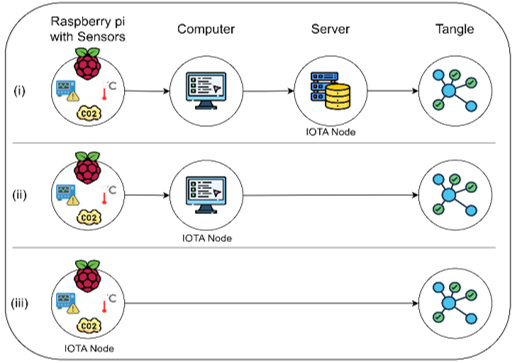
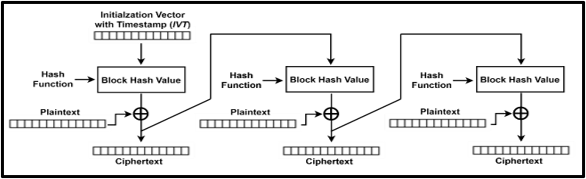
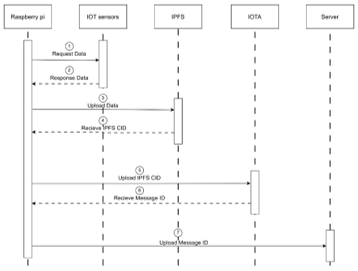
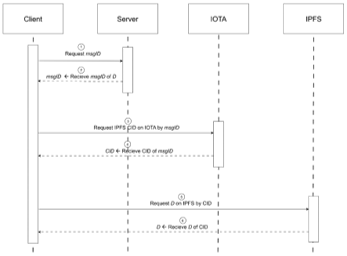
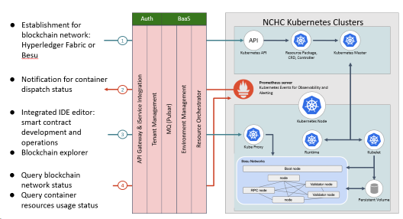
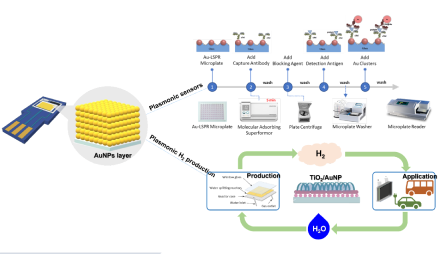
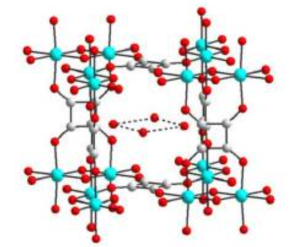
Devices and processing methods for capturing and utilizing carbon dioxide
Prof. Ming-Chun Lu / Department of Environmental Engineering
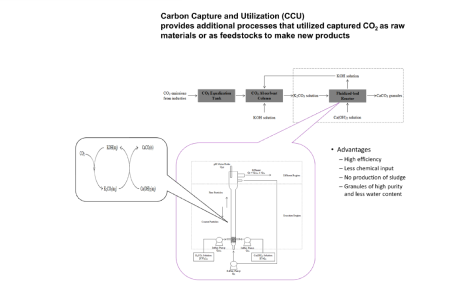
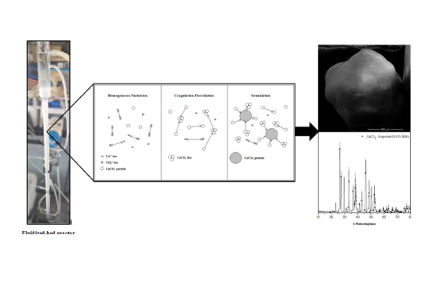
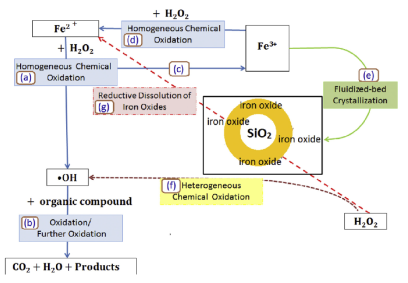
The technology developed by Professor Ming-Chun Lu to capture carbon dioxide from the flue gas with alkali at normal temperature and pressure, and then introduce it into the fluidized bed crystallization tank to produce calcium carbonate granules with calcium ions. The high-quality calcium carbonate crystals can be reused as additives in various production processes. This technology has excellent potential in terms of zero emission and sustainable development.
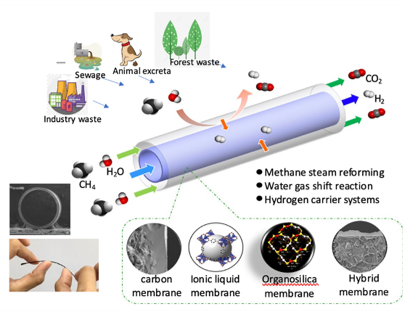
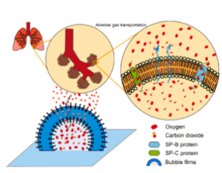
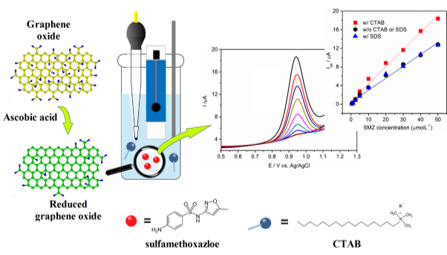
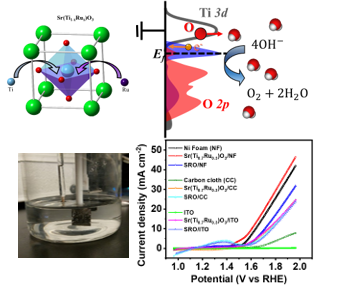
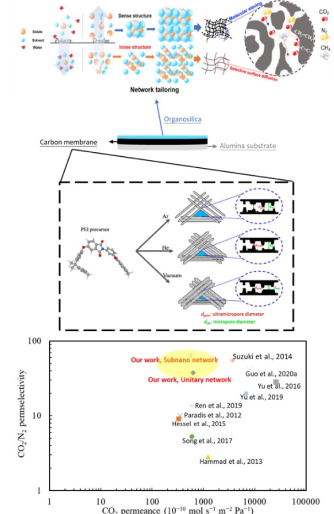
Development of inorganic carbon/silica composite membrane with CO2 facilitated transportation
Prof. Hui-Hsin (Anna) Tseng / Department of Environmental Engineering
Carbon/silica composite membrane developed by the research team of Prof. Hui-Hsin (Anna) Tseng
- Top layer is organosilica material with strong CO2-phlic property,which improves the CO2 suface diffusion and therefore boost the transportation of carbon dioxide
- Bottom layer is carbon molecular sieving membrane,possessing the angstrom-level pore structure, which is close to the size of gases molecule, and thus demonstrating the outstanding separation capability
- Taking advantage of the combination of both membrane is not only to provide selective gas pathwaybut also to facilitate CO2 transportation
- Compared with other gas separation membranes, this one shows excellent separation performance.
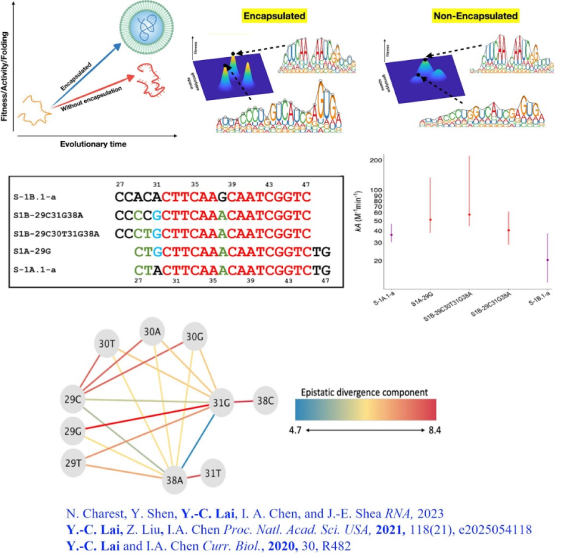
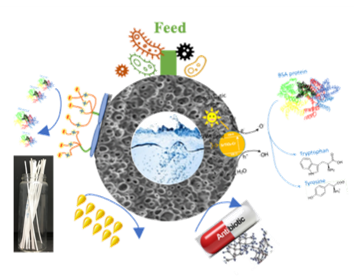
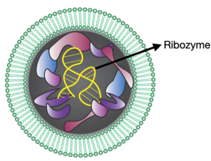
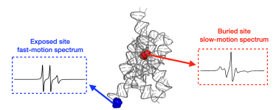
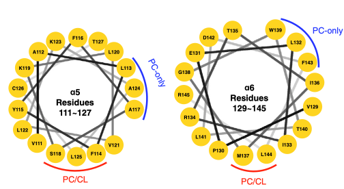
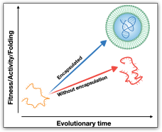
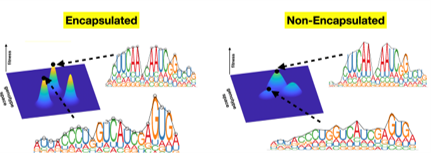
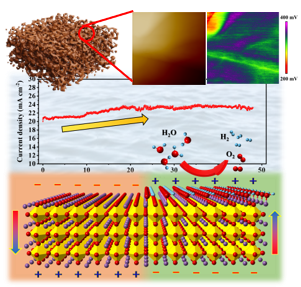
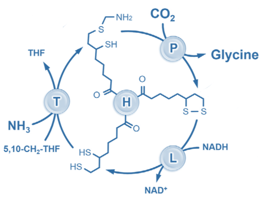
Carbon circulation and molecular evolution in the RNA world
Prof. Yei-Chen Lai / Department of Chemistry
Our research provides insights into carbon circulation and molecular evolution within the context of the RNA world, focusing on the interplay between protocells and ribozymes.
Our study demonstrates how ribozymes, when being encapsulated in protocells, significantly enhance their self-aminoacylation activity, a key step of carbon cycling and molecular evolution. We show that encapsulation offers a competitive advantage in prebiotic environments in fostering rapid evolutionary adaptation. The result underscores the importance of protocellular environments in ribozyme evolution (Results were published in Proc. Natl. Acad. Sci. USA).
We also utilize the information theory to extend beyond experimental constraints in identifying and predicting catalytic RNA sequences. The developed predictive model successfully identifies high-activity ribozyme mutants, which is crucial for understanding molecular evolution pathways (Results were published in RNA). Our result sheds light on the complex mechanisms of carbon circulation and molecular evolution in the RNA world, enhancing our understanding of the origins and functionality of RNA in early life forms.
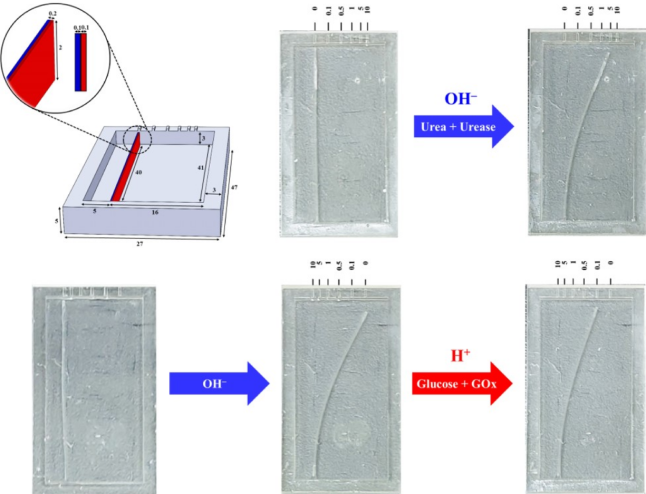
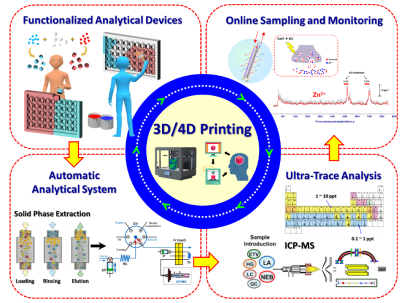
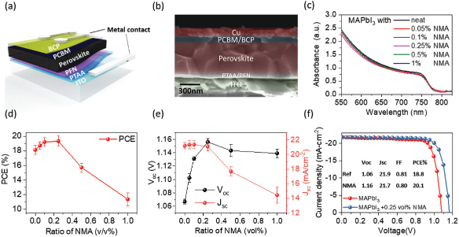
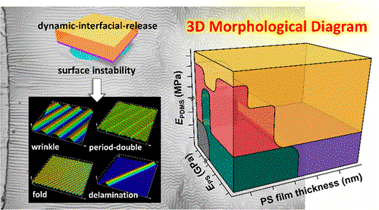
4D-Printed Unpowered Devices for Quantitative Chemical Analysis
Prof. Cheng-Kuan Su / Department of Chemistry
Our research team has made a groundbreaking development in utilizing four-dimensional printing (4DP) technologies for the direct fabrication of unpowered stimuli-responsive analytical devices for quantitative chemical analysis. We used digital light processing three-dimensional printing (3DP) and [H+]-responsive photocurable resins to fabricate all-in-one needle panel meters, with the method’s detection limits of 4.9 and 7.0 μM for urea and glucose, respectively. We verified the reliability of this analytical method by determining the concentrations of urea and glucose in samples of human urine, fetal bovine serum, and rat plasma and comparing the results with those obtained using commercial assay kits. Our results suggest that 4DP technologies can allow the direct fabrication of unpowered stimuli-responsive analytical devices for on-site quantitative chemical analysis and advance the applicability of 3DP-enabling analytical methods. (The results have been published in Biosensors and Bioelectronics)

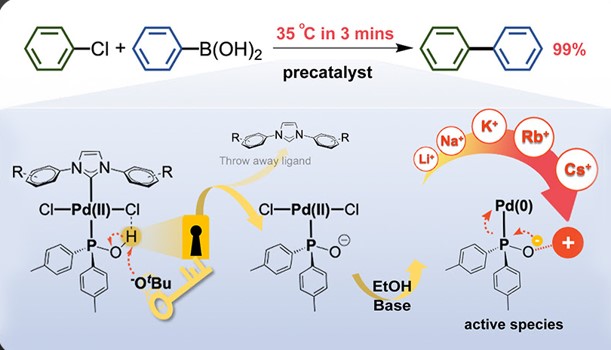
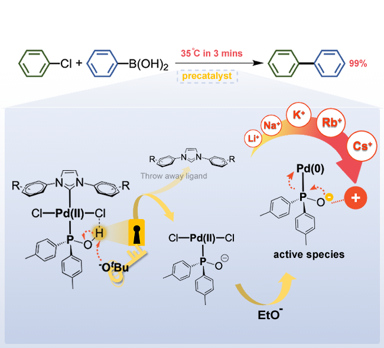
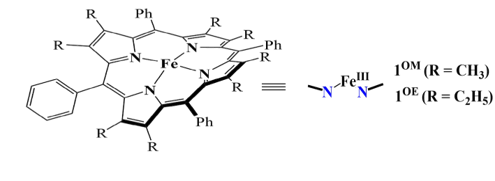
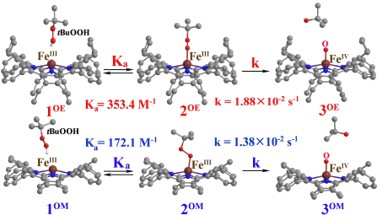
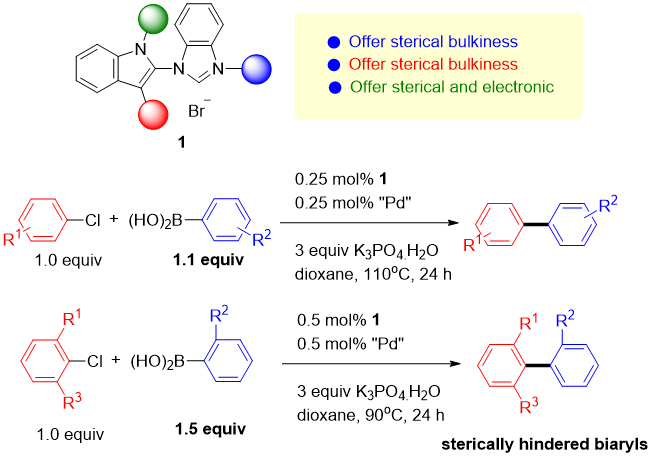
Development of Next-Generation Palladium Pre-Catalysts
Prof. I-Chung Lu / Department of Chemistry
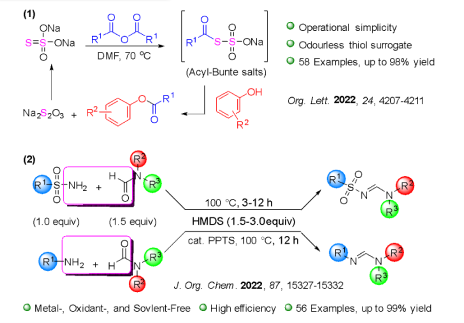
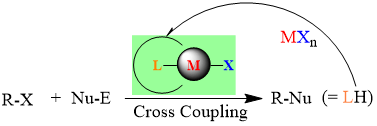
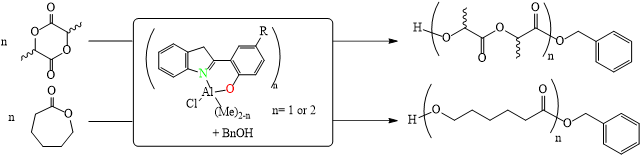
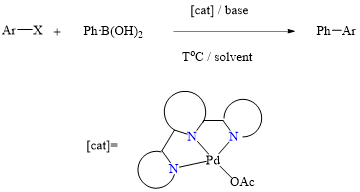
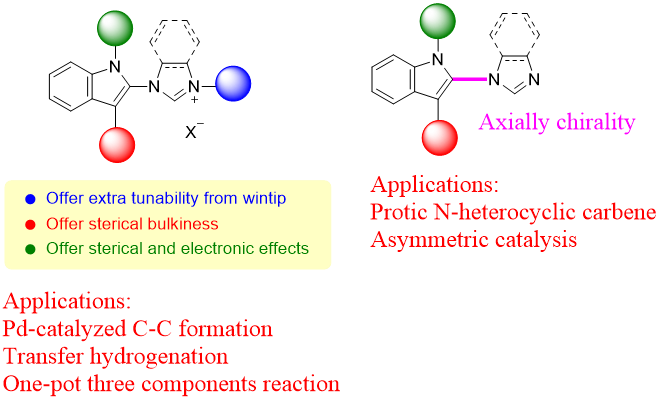
Our research team, in collaboration with Professor Chang, has successfully developed a highly efficient palladium pre-catalyst. This catalyst facilitates the carbon-carbon bond coupling reaction of low-reactive chlorobenzene in just three minutes at 35°C, achieving an impressive yield of 99%. The catalytic active species involved in this process are unique, deviating significantly from conventional reaction mechanisms. This opens avenues for chemists to regulate the activity using alkali metal cations and the basicity of anions in salt additives (published in ChemCatChem).
This pre-catalyst is exceptionally stable and holds significant application value. It exhibits outstanding catalytic cross-coupling capabilities in various reaction systems, potentially replacing conventional catalysts to meet energy-saving goals in synthetic reactions.
4D-Printed Unpowered Devices for Quantitative Chemical Analysis
Prof. Cheng-Kuan Su / Department of Chemistry
Our research team has made a groundbreaking development in utilizing four-dimensional printing (4DP) technologies for the direct fabrication of unpowered stimuli-responsive analytical devices for quantitative chemical analysis. We used digital light processing three-dimensional printing (3DP) and [H+]-responsive photocurable resins to fabricate all-in-one needle panel meters, with the method’s detection limits of 4.9 and 7.0 μM for urea and glucose, respectively. We verified the reliability of this analytical method by determining the concentrations of urea and glucose in samples of human urine, fetal bovine serum, and rat plasma and comparing the results with those obtained using commercial assay kits. Our results suggest that 4DP technologies can allow the direct fabrication of unpowered stimuli-responsive analytical devices for on-site quantitative chemical analysis and advance the applicability of 3DP-enabling analytical methods. (The results have been published in Biosensors and Bioelectronics)